The customizable bioinformatics system designed to accelerate your research
BRICS, an enterprise solution, is a collaborative and extensible web-based system to support the collection of research studies and clinical trials, using a set of modular components that cover all stages of the research life cycle. Importantly it's also a comprehensive integrated, end-to-end platform for clinical trial management, which allow investigators to design, conduct and manage studies easily and securely. And because BRICS is un-branded and un-associated with a particular disease or organization, it can be customized to meet your research objectives. This web-based application is 21 CRF Part 11 compliant, secure, and intuitive to use. In addition, users with programming expertise can use an application programming interface (API) for more dynamic data analysis including Artificial Intelligence and Machine Learning (AI/ML) applications. BRICS is actively used across the NIH and by the DoD for data warehousing, and clinical trials research.
BRICS: The Software - Plug and play components for the entire research life cycle
BRICS offers researchers a secure platform and a suite of tools to promote standardization, communication, and collaboration across the research community and a data repository to hold genetic, phenotypic, clinical, and medical imaging data. These plug-and-play modules can be shared across disease categories or deployed and branded independently, depending on the needs of your program. Together, they provide a combination of web-based functionality and downloadable tools that support data definition, data contribution, and data access throughout the research life cycle.

Account Management
This is the application for creating, approving, and managing user accounts, including management of access controls, roles, permissions groups, and authorization to other BRICS modules. The traditional username/password Log in flow for BRICS has been replaced by NIH's Researcher Auth Service (RAS). This change will require all users to follow a set of steps to Log in/sign up for RAS and link your BRICS account to your RAS account. RAS supports 3 identity providers, a NIH PIV/CAC card, an account with Login.gov, or an ID.me account. If you have a NIH PIV/CAC card please use it, otherwise, please use Login.gov or ID.me.
Clinical Trials Management System (CTMS)
The Clinical Trials Management System (CTMS) provides critical support for the planning, and execution of clinical trials, provides reporting functions and management of important study documentation, correspondence, compliance and milestones. The CTMS provides users with a centralized location that is accessible for the storing and retrieval of study documents, with versioning. The CTMS tool integrates with the NIH PROTRAK system and is able to import, IRB documentation, milestones, dates and many other critical study information seamlessly.

Data Dictionary: Data Elements, eFORMS, Form Structures
The BRICS data dictionary is a well-designed and intelligent tool, created in close collaboration with key projects and organizations such as the NINDS CDE project, NIH CDE repository, CDISC, LOINC, FHIR, UMLS , and other controlled vocabularies/terminologies. It is equipped with terminology and services that ensure compliance with various vocabulary standards, enabling the seamless exchange of clinical research data in diverse formats across systems and sources.
By embedding the BRICS data dictionary into informatics systems, users can promote the FAIR principles (Findability, Accessibility, Interoperability, and Reusability) and benefit from:
- Standardization: Support for standardized clinical research data across different projects through the incorporation of controlled vocabularies.
- Data Clarity: Structured descriptions of clinical research data that remove ambiguities.
- Data Discovery and Reporting: Enhanced ability to find, query, and report data efficiently.
- Interoperability: Facilitation of data exchange and comparison across independent informatics systems.
- BRICS data dictionary supports the conversion of CDEs to REDCap format.
OMOP (Observational Medical Outcomes Partnership)
The BRICS data dictionary includes tools that support the conversion of Common Data Elements (CDEs) into OMOP format. This facilitates the integration of research data with the OMOP common data model, enabling researchers to leverage standardized observational data for large-scale analysis and insights.
FHIR (Fast Healthcare Interoperability Resources)-to-CDE Tool
The data dictionary includes a FHIR-to-CDEs tool that ensures compatibility between clinical research data and FHIR profiles. This mapping enables the seamless integration of clinical data collected via FHIR-compliant systems into the BRICS ecosystem, ensuring a streamlined flow of standardized data between healthcare and research systems.
CDISC-to-CDE Tool
The BRICS Data Dictionary also includes a CDISC-to-CDE tool, which implements the Study Data Tabulation Model Implementation Guide (SDTMIG) to guide users on the organization, structure, and format of standard clinical study tabulation datasets, using the BRICS Platform. This also enables BRICS to support seamless data exchange between SDTMIG-compliant systems and BRICS, or submission to a regulatory authority, such as the US Food and Drug Administration (FDA).
UMLS (Unified Medical Language System)
The BRICS data dictionary incorporates a mapping tool that links UMLS concepts to BRICS CDEs. This integration allows users to leverage UMLS’s vast repository of medical terminologies and concepts, ensuring alignment between clinical research data and standard medical terminologies, further enhancing data interoperability and accuracy.
Launch Data Element to UMLS Mapping Tool
AIMS4DE: Artificial Intelligence Meaningful Search for Data Elements (Beta Version)
The FITBIR Instance of the BRICS data dictionary includes a Beta version of an AI semantic (meaningful) search tool for cross-mapping new study variables to previously used and standardized data elements archived in the FITBIR data dictionary. The semantic search tool employs a large language model to convert text to a numerical representation, so unlike a traditional keyword-matching search, this algorithm enables the user to search the data element dictionary based on the phrase’s meaning, regardless of spelling. Note that this is a beta version of the tool, which is currently only available for the FITBIR BRICS instance. The BRICS team is working to incorporate this tool more seamlessly into the current data dictionary search interface and has plans to potentially release similar tools for other BRICS instances. If you have questions, comments, or suggestions regarding your user experience or research needs, please don’t hesitate to contact maria.bagonis@nih.gov.
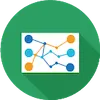
Data Mapping And Transformation (DMT) Tool
The Data Mapping and Transformation (DMT) tool is an application used to translate data from users’ systems, that may not have consistently used BRICS CDEs, to CDEs used in the BRICS system. In addition, the system translation tool saves the translated data into a BRICS compliant CSV that can then processed by the Validation tool.

Data Repository: Study Management, Data Validation And Submission
The Data Repository is the central hub of the BRICS system, providing functionality for defining and managing study information, and for contributing, uploading, and storing the research data associated with each study. In the pursuit of making information discoverable for the research communities that the BRICS system serves, the following attributes have been incorporated into the Data Repository. ORCID identifier (ORCID iD) is a unique, personal, persistent identifier for researchers that distinguishes them from every other researcher. The BRICS system allows a researcher to include their ORCID iD. The Office of Science and Technical Information (OSTI) maintains a ** DOI ** minting service called the "DOE Data ID Service." Each BRICS study, using the OSTI DOI minting service (DataCite) can create a unique DOI. In addition, the BRICS system has implemented the Data Tag Suite (DATS) model to support the DataMed data discovery index with the goal to be for data what PubMed has been for the scientific literature.

Forum
Forum is a discussion board for account users for posting messages, interacting with each other, and discussing various topics. It is organized into categories or topics, with each thread containing posts and replies from multiple users. It is moderated by operations team members to ensure that the posts and comments have appropriate content. This is currently enabled on FITBIR (on both Portal and Public Sites), cdRNS (on both Portal and Public Sites) and NEI (only on Portal).

GUID: Global Unique Identifier - A Privacy Preserving Record Linkage System
The GUID is a Global Unique Identifier for each study participant that allows researchers to aggregate and share a participant’s data without exposing personally identifiable information (PII). The GUID is made up of random alpha-numeric characters and is not generated from PII/PHI.[1]
[1] Johnson SB, Whitney G, McAuliffe M, Wang H, McCreedy E, Leon Rozenblit L, Evans CC. Using Global Unique Identifiers to Link Autism Collections. J. Am. Med. Inform. Assoc., Vol. 17, No. 6, 689-695, 2010. PMCID: PMC3000750.
*Note: Since the time of publication, the term GUID has taken on a more general meaning – “An implementation of the universally unique ID”. In the context of the BRICS system, the GUID process could now be better identified as a Privacy Preserving Record Linkage (PPRL) system. However, since the BRICS GUID system has been using the GUID term in documentation and software for more than a decade and it is familiar to our community, we will continue to use it for now.

Imaging Tools: MIPAV Imaging Data Submission Tool
The MIPAV (Medical Image Processing, Analysis, and Visualization) application enables quantitative analysis and viewing of medical images, such as PET, MRI, CT, or microscopy. A MIPAV plugin is used to package and submit image data of many formats (i.e. DICOM, NIFTI, BIDS, Analyze, AFNI and many others) into BRICS.
"Focusing on open source components, MIPAV is the best candidate for 3D imaging as well as DICOM communication" (http://link.springer.com/article/10.1007%2Fs10278-015-9833-1)
For more information on MIPAV, visit https://mipav.cit.nih.gov/
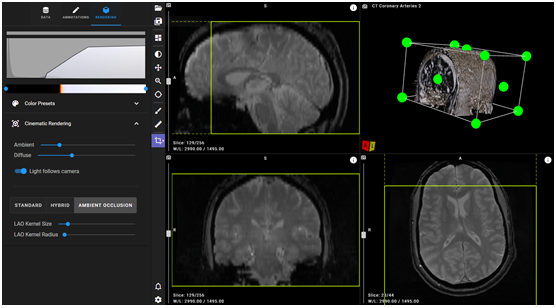
VolView in the BRICS Query Tool
BRICS integrates VolView into the Query Tool for users to view high-quality, interactive cinematic volume renderings and tri-planar visualization of the image volumes returned by their queries into the data repository. VolView is an open source radiological viewer developed for clinical professionals, offering 2D and 3D controls, measures, and annotations.
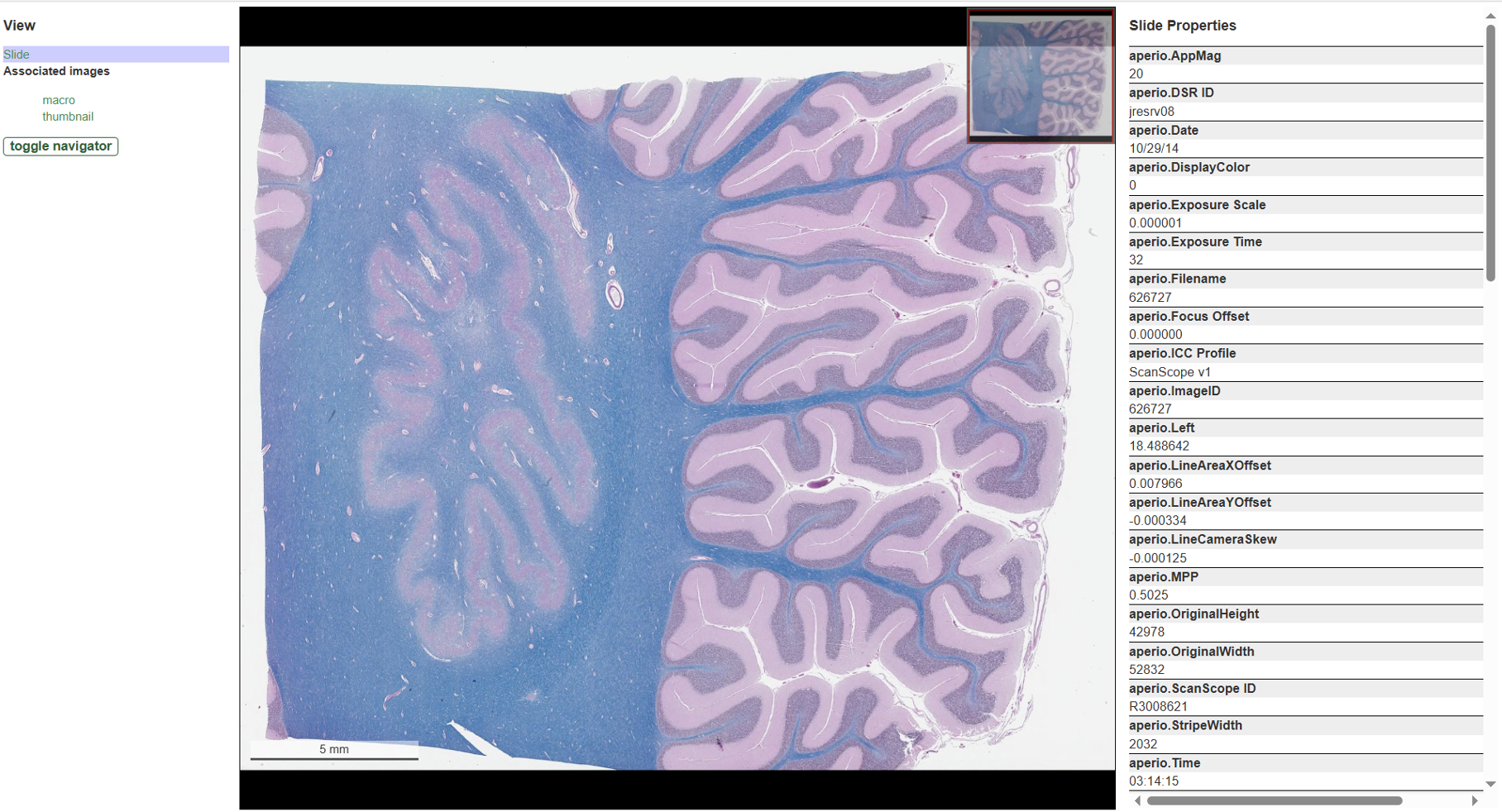
Virtual Slider Viewer
Virtual slide images are large, high-resolution images used in digital pathology. Displaying virtual slide images with standard image processing tools is a challenge due to their large size. The multi-resolution aspect of the virtual slide images is key to enabling the user to zoom into areas of interest in the image and see finer and finer details.
Investigator Education and Training (InET)
Research often requires the management and tracking of personnel trainings, licenses, and related documentation (i.e. CVs, on boarding, roles, position, credentials, etc.). Some items such as trainings and licenses have predefined validity periods and need to be updated. In addition, the process of onboarding new personnel, or offboarding existing personnel often involves documentation and the coordinated effort across the study team, PI and other groups such as study monitor etc. The goal of InET Is to provide an effective solution for the management of the various required study personnel trainings and documentation in a central location that is accessible. In addition, InET is able to generate lists of specific trainings based on role (Physicial vs Post bac) or function (clinical research with patient contact vs sample management), and InET is able to track document versions and notify users of pending items or expiring trainings or documents. InET helps to ensure that users are continuously in compliance with research and clinical trials.

Datastore
The BRICS Datastore module is a flexible data store allowing for management and storage of primary and secondary data from research studies. Each Datastore is assigned a unique persistent identifier (a digital object identifier (DOI)) to support data discovery and provides controlled access (permission based) to datasets and metadata which coexist within the module. With the new NIH Data Management and Sharing Policy coming into effect January 25, 2023, utilization of the BRICS Datastore module aligns with NIH’s mandate to use a quality data repository that improves the FAIRness (Findable, Accessible, Interoperable, and Re-usable) of the data.
The two main use cases for the BRICS Datastore module are as follows:
- Meta Analyses
- Facilitates aggregation of data from other studies within the BRICS Repository module for meta analyses
- Facilitates aggregation of data from other studies within the BRICS Repository plus accommodates upload(s) of data external to BRICS to be included in meta analyses - Data Store/Repository
- Facilitates storage of data from studies that do not have requirements to upload data to the BRICS Repository module
- Supports NIH Data Management and Sharing Policy

ProFoRMS: Protocol Management And Data Capture
The ProFoRMS tool serves as the main module for electronic data capture, subject management, and scheduling. ProFoRMS’s electronic case reports forms (eCRFs) support both standard and custom eforms using the CDE library. In addition, this module includes support for the PROMIS API, empowering access to a state-of-the-art assessment system for self-reported & adaptive measures, which seamlessly integrate into the BRICS common semantic model.
Data can be collected remotely using a patient self-report module (PSR), or offline using the Offline System for Trials and Research Information Collection and Housing (OSTRICH) app (compatible with iOS, Android and Windows), a BRICS application that facilitates offline data collection and ingestion into ProFoRMS once online connectivity is available. OSTRICH is currently available for iOS devices in the Apple App store and for Android devices in the Google Play store. Please go to the respective app store and search OSTRICH data collection. Please install the app. When the app first launches, please select the instance that the user will be using (i.e. Production or Demo). To change instance in the future, please remove OSTRICH and reinstall. For the Windows version of OSTRICH, please contact the support team. The system also offers electronic consenting capabilities and features to allow anonymous survey collection, if needed. Moreover, ProFoRMS includes important functionality including: question skip logic, auto calculations, reporting, data validation and more.
As part of the 21 CFR Part 11 compliance, ProFoRMS has complete audit trails for data collection and inbuilt data auditing capabilities. Subsequently, electronic records and electronic signatures within BRICS are considered trustworthy, reliable, and equivalent to paper records.
The 21 CFR Part 11 System Documentations contains the following documents: System Design Document, E-Signature Security Policy, Master Test Plan, Report on 21 CFR Part 11 Compliance, Software Development Process, System Requirements Specification, System Access Logs and User Guidelines. ProFoRMS also has the capability to integrate with EHR system databases.
At the NIH, this system is integrated with BTRIS and researches are able to obtain clinical data, seamlessly. In addition, FHIR connectivity is supported.
Additional Functionality:
- Digitized versions of common protocol logs (monitoring visits, delegation of authority, screening – enrollment, subject identification, training, deviations, UADE, UP)
- E-binder is available with audit trail
- System can support multi-site clinical trials and research protocols
- There is full role-based control for access to the system’s features
- Raw data export can be filtered and accessed in real-time – helpful for DSMB reporting.
- Anonymous survey capabilities – remote data collection
- Ability to perform randomization of subjects, blind/unblind users, control randomization based on eligibility criteria

Query And Export Data
The Query Tool provides access to harmonized data submitted by researchers. The efficiency of the application sifts through data by filtering on data elements and form structures. Moreover, Boolean operators (AND, NOT, OR), specificity (inclusive and exclusive), and joins across multiple form structures provide additional data exploration. The Query Tool API is an extension of the capabilities of this powerful tool which allows users to access data (JSON or CSV format) using their preferred IDE, such a Jupyter notebooks, and programming languages such as R and Python. The API allows researchers/data scientist to write Python code, incorporating Python’s extensive AI/ML libraries/ecosystem to provide AI/ML data analysis. Importantly, since the BRICS data dictionary/CDEs are a foundational aspect of BRICS, the CDEs provide accurate and rich metadata for each data point furthering the programmatic AI/ML analysis on both on-premise and cloud instances of BRICS.
- Query User Guide
- BRICS API User Guide
API Scripts (Zipped)
api-scripts.zip (1.84 MB)BRICS API UserGuide Notebook File (Zipped)
brics_api_userguide.zip (580.98 KB)FITBIR GOSE Notebook File (Zipped)
fitbir_gose.zip (19.61 KB)- GitHub Link: https://github.com/brics-dev/data-discovery-api
STAMS: Specimen Tracking And Management System
STAMS is a comprehensive application for the tracking and management of samples that provides the ability to record complete sample information, including sample history and can be easily adapted to support a number of different sample types. STAMS pro vides the options for physical freezer and container inventory management and storage organization, and can be customized to match the physical dimensions of the freezer, tray and storage box. The integration with commercial barcode scanners and barcode printers facilities efficiencies with biosample collection, storage, management and retrieval. STAMS also has an intergrated brain pathology tool that is specifically designed for the management of brain repositories to include the ability for clinical recording of stains, brain slice annotation, next of kin interviews and the generation of pathologist reports. STAMS is integrated with the NIH medical records system via BTRIS and is able to ingest and display patient medical record information, and also has active support for ICD diagnosis codes.
STAMS Capabilities:
- Any sample type (fluid, tissue etc.)
- Bar codes can be printed from within the system (currently Zebra printers are supported)
- Bar codes can be scanned directly into the system
- Samples can be linked to subject information in ProFoRMS
- Sample quantities and use can be tracked and monitored
- Ability to retrieve subject data (imaging, clinical data, and samples) in a comprehensive manner.